CaseCryo® NON-DMSO
Hot







CaseCryo® NON-DMSO
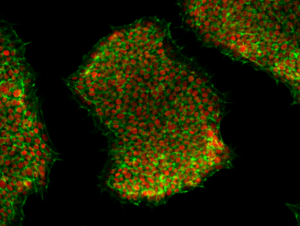
CaseCryo® NON-DMSO is a chemically defined, completely animal component-free and protein-free freezing medium with optimized alternative cryoprotectants eliminating the need for DMSO. This medium has been specially formulated and empirically tested for the cryopreservation of human pluripotent stem cells (hPSCs) and other human cell types. In direct comparisons to other commercially available DMSO-free freezing media, CaseCryo® NON-DMSO displays vastly superior viable cell recovery and long-term cell function and expansion. CaseCryo® NON-DMSO is also serum-free and animal-free which reduces immunogenicity providing a safer alternative to other freezing media. CaseCryo® NON-DMSO has been optimized for hPSCs, but it is versatile and displays excellent recovery of cell types derived from hPSCs and other human cells including HEK293 cells. This product can be used synergistically with CaseBase™ Dissociation Medium and CaseBase™ Washing Medium to improve cell survival and function in cryopreservation workflows.
All of CaseBioscience media, classical media, and balanced salt solutions are manufactured in our advanced, cGMP, ISO 13485:2016 certified facilities. With stringent adherence to the highest quality and consistency in manufacturing and testing parameters, CaseBioscience prioritizes performance and reproducibility in your biological research and downstream processes.
Join Our Mailing List
Be the first to know about new products, special events, and more!
